 HL7 CCPM, R1 HL7 Version 3 Standard: Common Product Model CMETs, DSTU Release 11 June 2011 |
Content Last Edited: 2011-06-14T14:26:17
3.2 Storyboards
3.3 Refined Message Information Models
3.4 Hierarchical Message Descriptions
Individual product instances (with serial numbers), or product lots (with lot numbers.)
|
||||||
|
For details on the interpretation of this section, see the storyboard discussion in the Version 3 Guide.
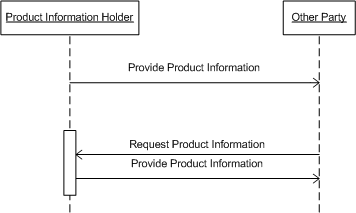
The following storyboards reflect the use of products in the ICSR arena
Mrs. Nuclear, an 86 year old female consumed 400 IU of vitamin D made by Supplement Company IOP. Four hours later she fell to the floor and had a seizure. When she stopped seizing, her husband noted that she did not awake, and called the 911-Emergency Dispatcher. When the paramedics arrived, Mrs. Nuclear was still unconscious and she was taken to the hospital. When she arrived at the hospital, Mrs. Nuclear regained consciousness in the examination room, and the emergency room attending physician, Dr. Attend ordered a series of tests to be performed to obtain a more precise diagnosis of Mrs. Nuclear condition. Mr. Nuclear informed the attending physician that they had started taking a new vitamin D supplement, and that he suspects that Mrs. Nuclear is having an adverse reaction to the supplement. Dr. Attend responded to Mr. Nuclear that he does not suspect this to be the case, but informs Mr. Nuclear that he can report the incident to the dietary supplement company or FDA to help alleviate his suspicion. Mr. Nuclear called the FDA's Consumer Complaint hotline to report the problem, and provided a contact number for Dr. Attend for follow up once the laboratory results are final. The FDA Consumer Complaint hotline technician data entered the complaint into the system, and sent the case electronically to the FDA Center for Food and Applied Nutrition (CFSAN) for further analysis and follow up.
Great Cracker Company's Plant in St Cloud, MN produces two box sizes of Great Crackers--a 12 ounce box and a family-size 18 ounce box. On July 3rd, 2008, a routine QA inspection of lots produced from July 1st through July 3rd detected a strange odor during a tasting of samples from each box. Upon further testing, it was determined that the products had been contaminated with a very deadly pesticide. The quantity of production during this Time frame was 10,000 boxes of the 12 ounce size (5,000 in lot # 2008-0701-200, and 5,000 in lot # 2008-0702-201) and 8,000 boxes of the 18 ounce size Box (lot # 2008-0703-202).
Before the problem was discovered, 6,000 of the 12-ounce (5,000 from lot # 2008-0701-200 and 1,000 from the other lot) and 6,000 of the 18-ounce Boxes were shipped to a local grocery warehouse in St Paul and 1,000 of the 18-ounce boxes were shipped to another local grocery warehouse in Richmond, VA. It was determined that the adulteration was unintentional and happened at the St Cloud facility and was not due to adulterated raw materials from suppliers.
The two retail warehouses were notified on 3 July in time for the warehouses to quarantine the reportable food products.
The remaining Great Crackers were disposed of on 3 July at the St Cloud plant.
The company's risk manager and (the person who is in the FDA food facility registration database for the plant) logs into a Federal AE Portal and enters this report the evening of 3 July in compliance with 1005 legislation. His report includes the following information:
- Date food product is reportable on 3 July 2008
- The risk manager validates his FFRN and reports that the plant in St Cloud, MN is both the responsible party and the reporting site.
- Two (2) different products (12- and 18-ounce size boxes) the amount of the 12 ounce adulterated product--in both lots--was reported as was the lot amount of the 18 ounce product.
- The adulteration discovery code is 'self-discovery'.
- Both destinations of the shipments were identified and the amount shipped, by lot, to each location was reported.
- The risk manager reports that he has informed both 'down' locations of the adulteration on 3 July.
- There is no requirement for the company or the risk manager to report the origin of the raw materials, since the adulteration only took place in St Cloud.
- There is no requirement for either destination warehouse to report since they did not further distribute the reportable food prior to the notification and it was quarantined on site.
- Amount of adulterated food produced :
- Product 1 (12-ounce box; lot # 2008-0701-200) = package type = box; size = 12 ounces; package quantity = 5,000
- Product 1 (12-ounce box; lot # 2008-0701-201) = package type = box; size = 12 ounces; package quantity = 5,000
- Product 2 (18-ounce box; lot # 2008-0701-202) = package type = box; size = 18 ounces; package quantity = 8,000
- Amount disposed of:
- Product 1 (12 ounce box; lot # 2008-0701-201) - package type = box; size = 12 ounces; package quantity = 4,000
- Product 2 (18-ounce box; lot # 2008-0701-202) - package type = box; size = 18 ounces; package quantity = 2,000
- Amount shipped:
- To St Paul:
- Product 1 (12-ounce box; lot # 2008-0701-200) = package type = box; size = 12 ounces; package quantity = 5,000
- Product 1 (12-ounce box; lot # 2008-0701-201) = package type = box; size = 12 ounces; package quantity = 1,000
- To Richmond:
- Product 2 (18-ounce box; lot # 2008-0701-202) - package type = box; size = 18 ounces; package quantity = 6,000
- To St Paul:
Pharmacist Rebecca Rogers is preparing a solution to be used in chemotherapy for patients in General Hospital's outpatient cancer clinic. She notices, as she holds up a bottle from storage, that the solution appears cloudy and has particles floating in it. This physical property is not according to specification, and she sets the bottle aside. At the end of the day, a more thorough inspection reveals that half the bottles in a case of Solution X contain contaminated or defective solution. She reports the problem to the medical supply department, and the hospital files a product problem report with the regulatory authority and the manufacturer.
|
||||||||
|
For details on the interpretation of this section, see the description of RMIMs in the Version 3 Guide.
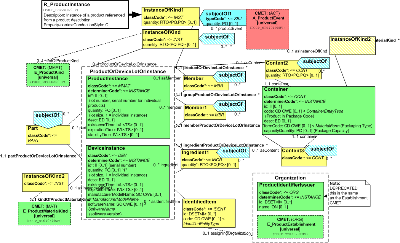
| Parent: | ProductKind (POCP_DM010000UV) |
Instance of a product referenced from a product desctiption.
| R_ProductInstance | POCP_HD020100UV01 |
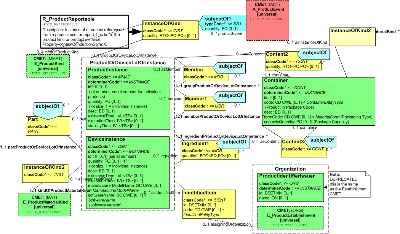
| Parent: | ProductKind (POCP_DM010000UV) |
Instance of a product referenced from a product use or report, has both, the product kind or product instance and either may be used.
| R_ProductReportable | POCP_HD020200UV01 |
|
||||||||
|
For details on the interpretation of this section, see the description of HMDs in the Version 3 Guide.
Instance of a product referenced from a product desctiption.
| E_ProductKindUniversal | POCP_MT010200UV01 |
| E_ProductMaterialKindUniversal | POCP_MT010400UV01 |
| E_ProductEstablishmentUniversal | POCP_MT030200UV01 |
| A_ProductEventUniversal | POCP_MT040100UV01 |
| R_ProductInstance | POCP_MT020100UV01 |
Instance of a product referenced from a product use or report, has both, the product kind or product instance and either may be used.
| E_ProductKindUniversal | POCP_MT010200UV01 |
| E_ProductMaterialKindUniversal | POCP_MT010400UV01 |
| E_ProductEstablishmentUniversal | POCP_MT030200UV01 |
| A_ProductEventUniversal | POCP_MT040100UV01 |
| R_ProductReporttable | POCP_MT020200UV01 |
| Return to top of page |

